Bioorganic Chemistry 87 (2019) 647–654
2019
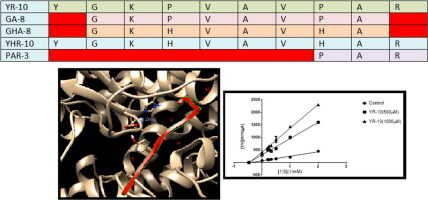
The structure-function relation of YR-10 (YGKPVAVPAR) was investigated by synthesizing four structural analogs of that including YHR-10 (YGKHVAVHAR), GA-8 (GKPVAVPA), GHA-8 (GKHVAVHA), and PAR-3 (PAR). GA-8 (GKPVAVPA) was synthesized on the basis of simulated enzymatic gastrointestinal digestion performed by bioinformatics tools (expasy-peptide cutter). This study explains the molecular mechanisms for the interaction of synthetic peptides with ACE. The IC50 values of each were 139.554 ± 2.3, 61.91 ± 1.2, 463.230 ± 3.56, 135.135 ± 2.1, 514.024 ± 5.86 µM, respectively. Results indicated that Pro replacement with His in YR-10 and GA-8 increased ACE inhibitory activity respectively, by 55.63% and 70.82%. Removal of Tyr and Arg from respectively N and C terminal positions of YR-10, following in silico simulated gastrointestinal digestion caused the 3.31 fold decrease in ACE inhibitory activity. YHR-10 showed the best docking poses, and GHA-8 exhibited interaction with Zn2+. Lineweaver–Burk plots of most active peptides suggest that they act as noncompetitive inhibitors against ACE.